Why trust Smart Cells?
Smart Cells is the best choice for giving your children a healthy future – and peace of mind.
The first private stem cell storage company in the UK, Smart Cells has been preserving and storing umbilical cord stem cells for more than twenty years. Smart Cells has successfully released more samples worldwide for treatment of life-threatening illnesses than any other private company in the UK. Now part of FamiCord Group, Europe’s largest stem cell bank, we bring expertise from more than 20 countries.
More than just a stem cell bank
Parents all around the world are choosing to store stem cells – there have been more than 700,000 cord blood collections globally. Smart Cells offer the best in technology, support, and customer experience to make this as easy as possible. All our samples released have been successfully used for treatment so far.
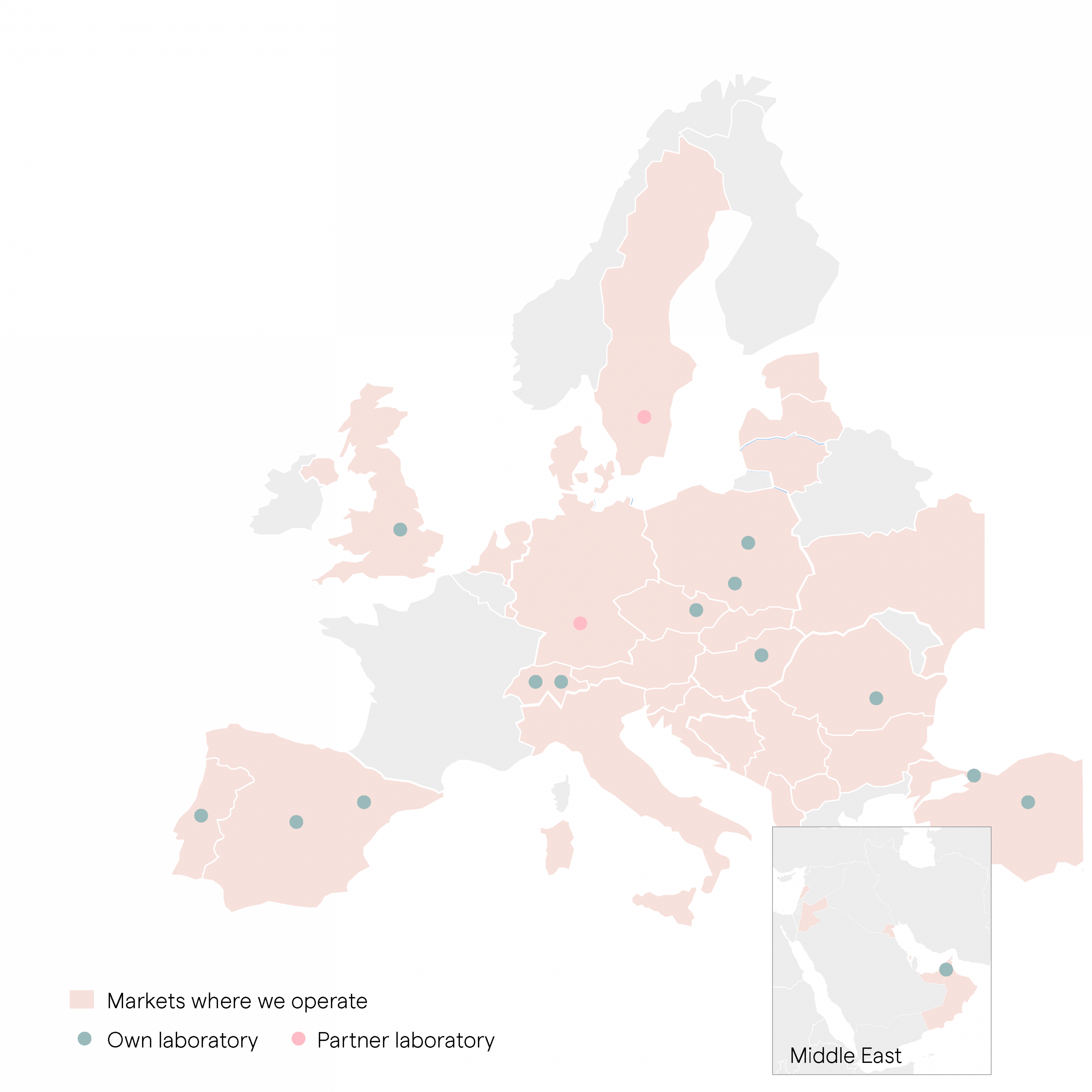
Now part of FamiCord Group, the largest cord blood bank in Europe and the third largest cord blood bank in the World. Our scale of operations is the best proof that we are serious about family umbilical cord blood banking and trusted by families all over the world.
#1 in Europe
Part of the FamiCord group, #3 in the world and present in 35 countries
Trusted by 500,000 families
With over 20 years’ experience
220+ Cord blood transplants
Stem cells delivered to 45+ countries worldwide
1,800+ Cord tissue therapies
Only family bank in Europe, of which the cord tissue and stem cells are used extensively in therapies
Leader in R&D
Providing personalised treatment for FamiCord Group clients
UK-based lab
Near Heathrow airport, with 12 other laboratories in Europe
Affordable storage
Flexible payment options and a price match guarantee
Smart Cells samples save lives
Smart Cells works closely with the best clinicians and maternity hospitals in the UK – they trust our research-led, quality approach above anyone else. A scientific director with over 35 years’ experience in the this field oversees the release of every sample, and ensures the highest levels of quality assurance.
Smart Cells care about the things you do
The team at Smart Cells care about your child’s future – and our planet’s. Smart Cells is proud to partner with leading health researchers such as the Borne project, and our supply chain partners are just as environmentally conscious as you are.
Cutting-edge technology, global reach
A state-of-the-art laboratory ensures samples are processed and stored using the latest science and technology. Samples are stored close to Heathrow airport, ensuring they can be collected and released from or to anywhere in the world – fast.
A dedicated UK phlebotomy service with emergency contacts and call out personnel operates 24/7 to save you the inconvenience of having to find, book, and manage this service yourself.
Exceptional customer service makes Smart Cells different
Smart Cells work with clients all around the world. The expert team supports you every step of the way. Pricing and service plans are clear and honest, as is our data on sample success – no misleading claims. All costs and guarantees are fully transparent from the start, including phlebotomy services (UK only), maternal blood testing, and shipping fees.
The team are happy to share data with you about how samples have been used for treatment.
Give your child the best chance possible in life. Our expert team can explain just how easy it is. They’re here to support you and talk through payment options.

Smart Cells Laboratory
The Smart Cells Laboratory uses state-of-the-art equipment designed to process and store cord blood as quickly and optimally as possible.
All processing procedures are undertaken in a sterile, controlled environment by specially trained technical staff, using the industry standard system for cord blood processing – the Biosafe Sepax system
Quality Assurance: samples are regularly tested and validated for sterility and to ensure required stem cell counts and viable cell recovery
The facility is alarmed and has an uninterruptible power supply in case of a mains electricity failure. The environment is continually monitored by an Advanced Facilities Management System.
Smart Cells holds a licence from the Human Tissue Authority
Accreditation
Our story
Our Founders – Shamshad and Fehmina Ahmed
While working as a managing director for a clinical trials company, Shamshad saw first-hand what stem cells could do to treat blood-related illnesses. Passionate about the possibilities stem cells offered for treating a range of illnesses, Shamshad founded Smart Cells International in 2000 to help parents store their baby’s umbilical blood stem cells for future use.
Smart Cells was the first private UK cord blood company, and has grown to become one of biggest in the UK, storing more than 50,000 cord blood samples from people in more than 70 countries. Shamshad’s goal to give parents access to potentially life-saving treatments for their family remains the same.


Shamshad Ahmed
Founder & CEO of Smart Cells International Ltd
‘’The number of samples we have released signifies that stem cell technology is one of the most exciting areas of medical science in our time. The fact that we have released more samples in the last ten years than we did in the first ten years shows this evolving area is making big steps in a positive direction.’’
FAQ’s
Smart Cells has an HTA licence which covers procurement, testing, processing, storage, import, release, and export of cord blood cells and tissue. Smart Cells has ISO9001:2015 certification, and participates in the UK National External Quality Assessment Service (NEQAS) quality assurance scheme for full blood and CD34 cell counting.
Smart Cells takes ongoing Quality Assurance very seriously, with a rigorous programme of validating and auditing all critical practices and procedures. Fully trained and experienced staff operate according to validated procedures. The Smart Cells laboratory technical team are educated to degree and post graduate degree level in relevant biomedical sciences and use globally recognised techniques and technologies in the state-of-the-art processing facility.
The Smart Cells approach to ongoing Quality Assurance, regulatory compliance, integrity, and transparency is supported and overseen by the Scientific Director, Head of Laboratory Operations, and Quality Officer.
With 28 years of experience in stem cell transplantation in the NHS, and considerable academic and research experience, Smart Cells’ Scientific Director Dr. Ann Smith ensures Smart Cells meets best practice in the field. Her expertise is available to parents, partners, and healthcare professionals directly or via the customer services team.
Establishments licensed by the HTA are legally required to ensure that in the event of activities ceasing, any tissues/cells and records are transferred to another suitably-licensed establishment. Smart Cells is compliant with this requirement.


