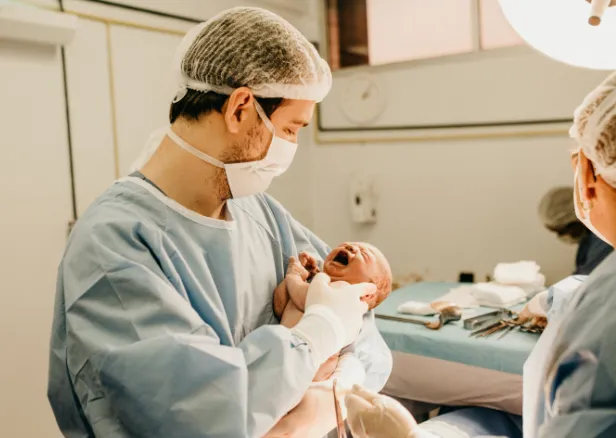
Our Laboratory
A state-of-the-art laboratory ensures samples are processed and stored using the latest science and technology.
Our laboratory uses state of the art equipment designed to process cord blood as quickly as possible. All processing procedures are undertaken in a sterile, controlled environment by specially trained staff. Samples are stored close to Heathrow airport, ensuring they can be collected and released from or to anywhere in the world – fast.
- We use the industry-standard system for cord blood processing
- The facility is based close to London Heathrow airport – minimising transportation times for customers around the world
- Our Advanced Facilities Management System continually monitors the environment; it’s alarmed and has an uninterruptible power supply in case of a mains electricity failure
- We hold a licence from the Human Tissue Authority

Smart Cells International (SCI) Quality Assurance
At SCI we take quality assurance very seriously and have a comprehensive and ongoing approach to validations and auditing of all critical processes and products. Our Quality Management System which applies to all activities undertaken by SCI, has been developed to meet the company’s high standards and is compliant with HTA regulations and ISO standards. Where possible, SCI aligns its practices and procedures with internationally recognised standards that apply to transplantation: NetCord-FACT International Standards for Cord Blood Collection, Banking, and Release for Administration and The Joint Accreditation Committee of the International Society for Cellular Therapy and the European Society for Blood and Marrow Transplantation (JACIE).

The laboratory and quality assurance:
We have a state of the art processing and storage laboratory which is monitored 24/7 and operated by trained and experienced technical staff. All samples collected on behalf of clients are fully traceable throughout their journey which includes testing, receipt, processing, cryopreservation, storage and release for treatment if required. Samples are safely stored in vapour phase nitrogen at temperatures so low that all the metabolic processes of the cells cease. Remarkably, these cells, if correctly handled, can be thawed and restored such that they can go on to grow and multiply in a patient undergoing transplant treatment.
We run validations to confirm that our processing laboratory consistently exceeds the standards of the current European Guide to Good Manufacturing Practice (GMP), Annex 1 of Directive 2003/94/EC. In this way, we can confirm that all samples are processed in ultra-clean conditions.
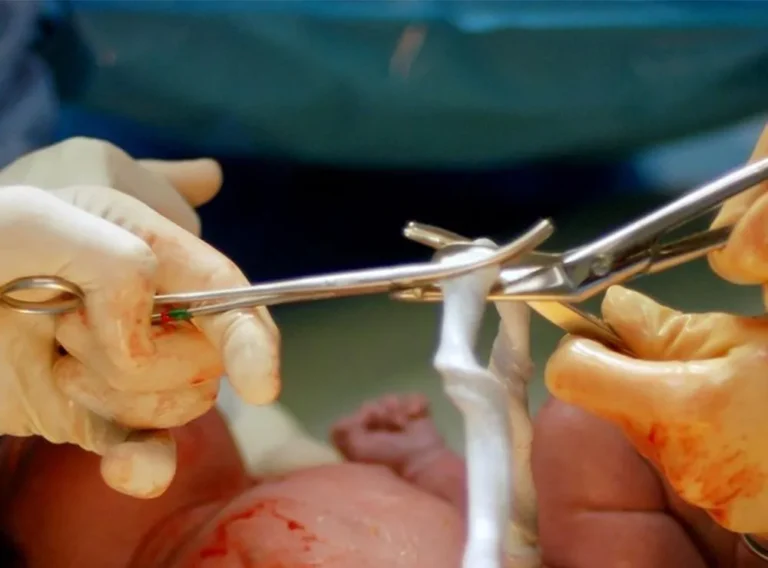
Every cord blood (CB) and cord tissue (CT) sample is transported to the laboratory as quickly as possible in approved, validated, temperature monitored transit containers.

Cord blood:
Following receipt and careful checking, all CB samples are tested for full blood and viable CD34 stem cell counts in-house. To ensure that our testing is consistently accurate we participate in independent UK External Quality Assurance Schemes (NEQAS). All samples are also tested for sterility by an accredited UK NHS laboratory. CB samples undergo skilled aseptic processing and storage in the laboratory. At SCI, we use the industry-standard system which is a functionally-closed technology developed specifically for processing cell therapy products. The system produces high quality, high yield volume reduced stem cell-enriched cord blood products for clinical use. This technology is used worldwide in both public and private cord blood banks and has a significant track record in the production of cellular therapy products that have been widely and successfully used in clinical practice.
To ensure good practice, we validate all our processing and cryopreservation procedures at least twice annually by thawing test aliquots to check cell numbers and viable cell recovery.

Cord tissue:
As far as CT samples are concerned, all undergo sterility testing in the same NHS laboratory that we use for CB testing. Following processing, several small test segments are reserved for each sample that we store. Representative batches of these are sent to an independent laboratory for viability testing, which checks for the presence of viable cells in the tissue. These validation tests are run at least twice per year and can also be arranged should a sample be proposed for use.
SCI presented and published details of their approach to validation of CT under varying transit conditions at the European Society for Blood and Marrow Transplantation annual meeting in 2016. (1 and 2)
(1). M.A. Smith et al. The effect of time and temperature during transportation of umbilical cord tissue on viability, functionality and differentiation potential of mesenchymal stem cells following cryopreservation and thawing. P038; S129 130. http://www.nature.com/articles/bmt201648
(2) View the poster presented here.

Rate this article: